1
/
of
12
www.ChineseStandard.us -- Field Test Asia Pte. Ltd.
YY/T 0471.5-2017 English PDF (YY/T0471.5-2017)
YY/T 0471.5-2017 English PDF (YY/T0471.5-2017)
Regular price
$170.00
Regular price
Sale price
$170.00
Unit price
/
per
Shipping calculated at checkout.
Couldn't load pickup availability
YY/T 0471.5-2017: Test Methods for Primary Wound Dressings - Part 5: Bacterial Barrier Properties
Delivery: 9 seconds. Download (and Email) true-PDF + Invoice.Get Quotation: Click YY/T 0471.5-2017 (Self-service in 1-minute)
Newer / historical versions: YY/T 0471.5-2017
Preview True-PDF
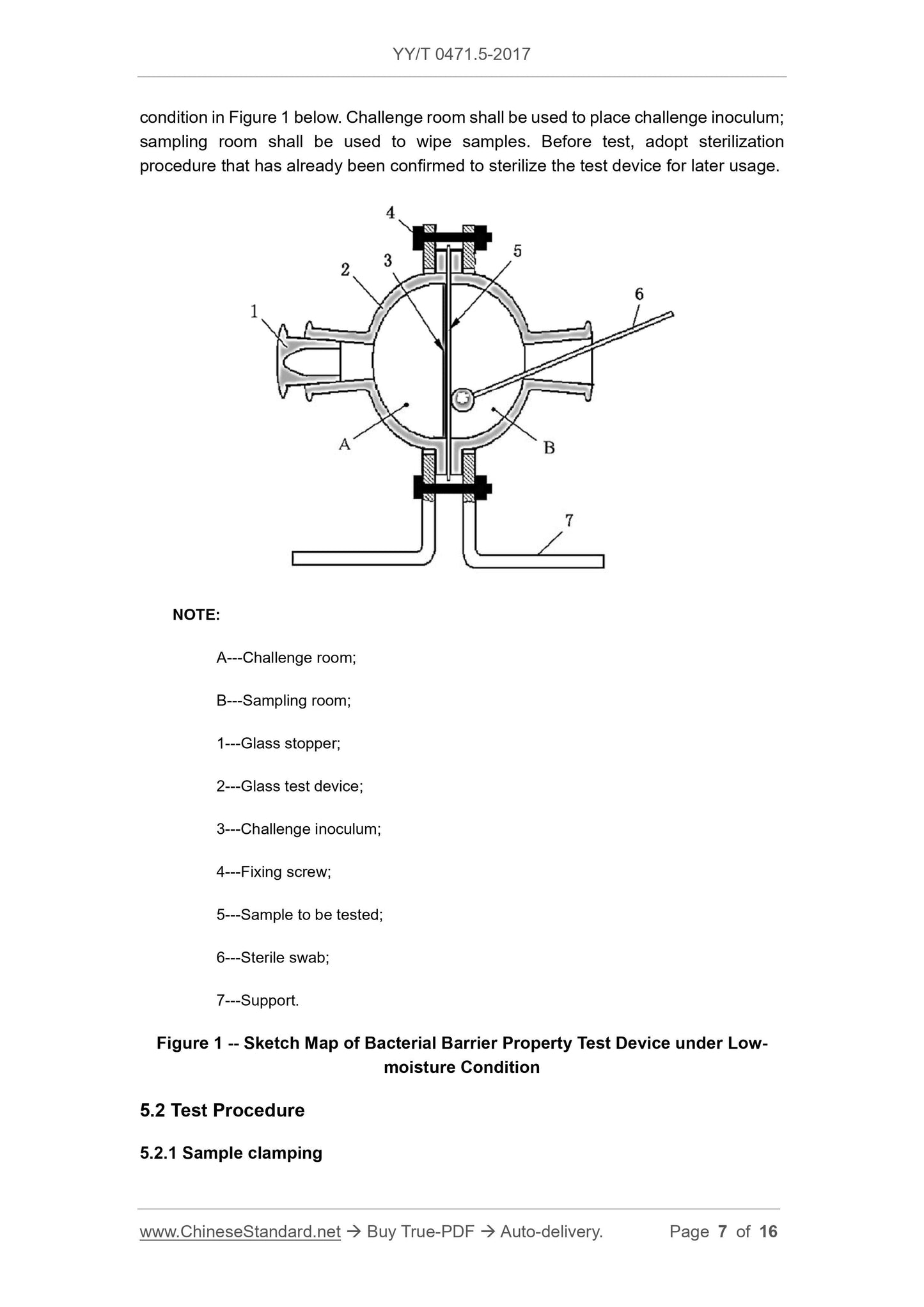
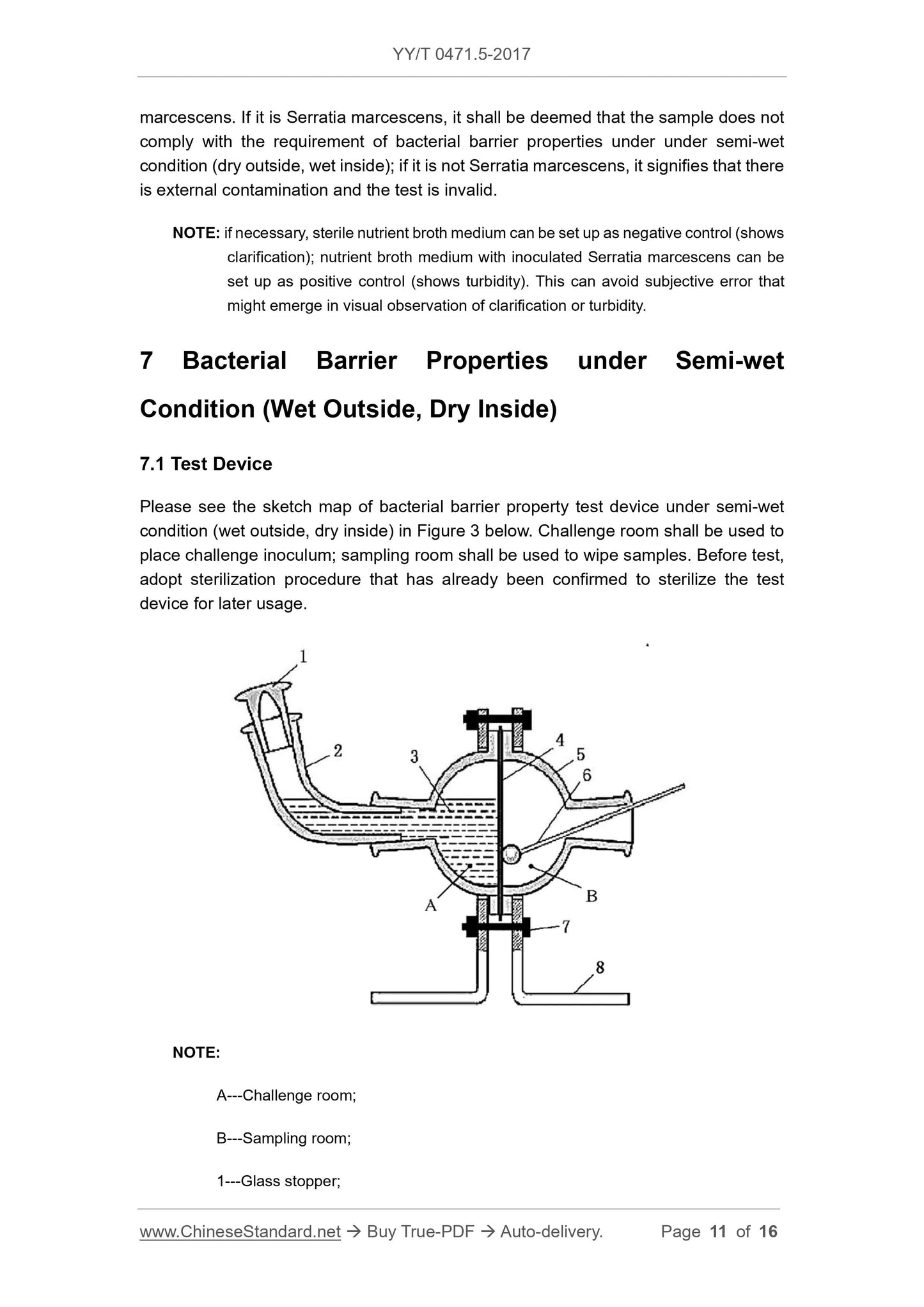
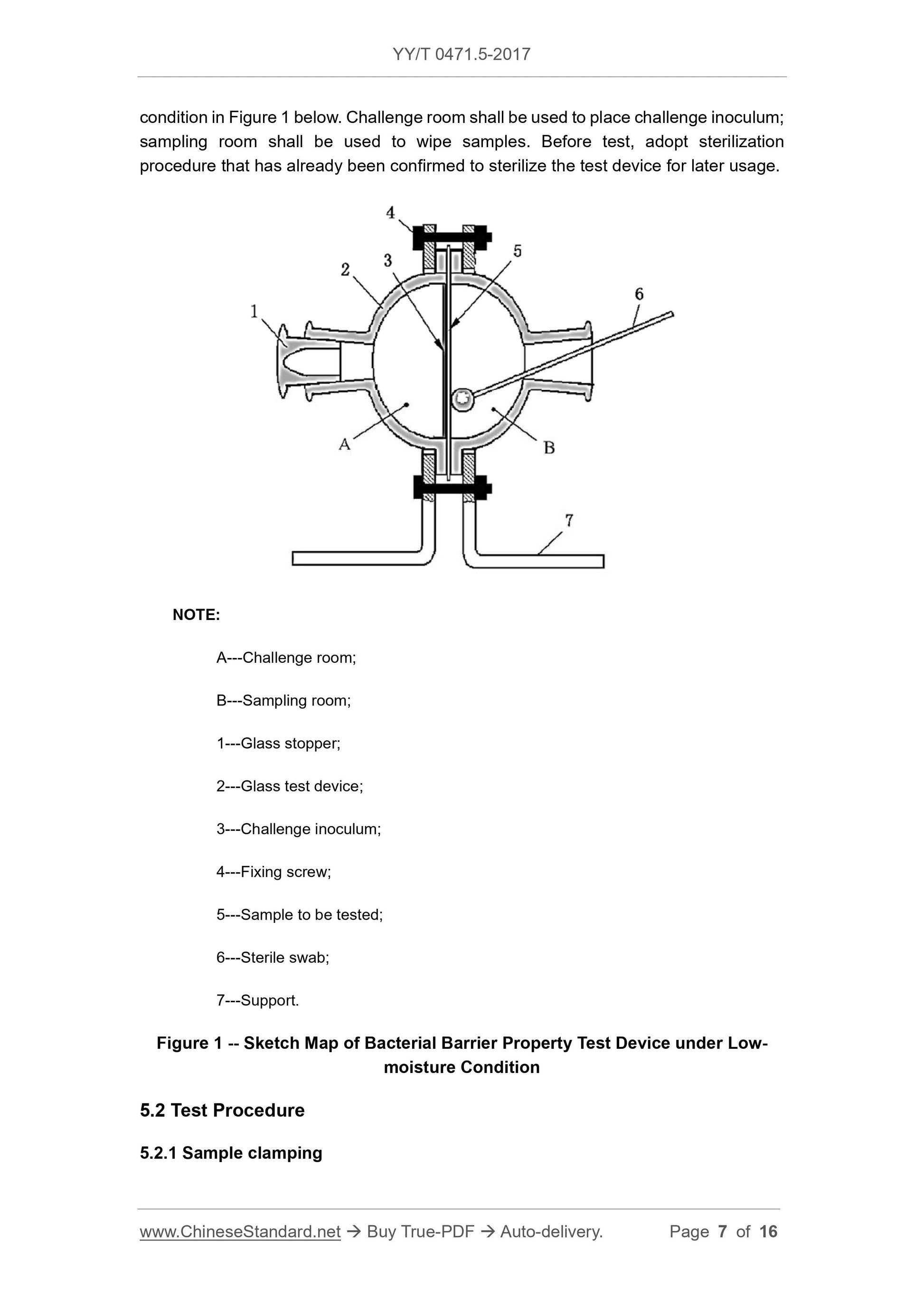
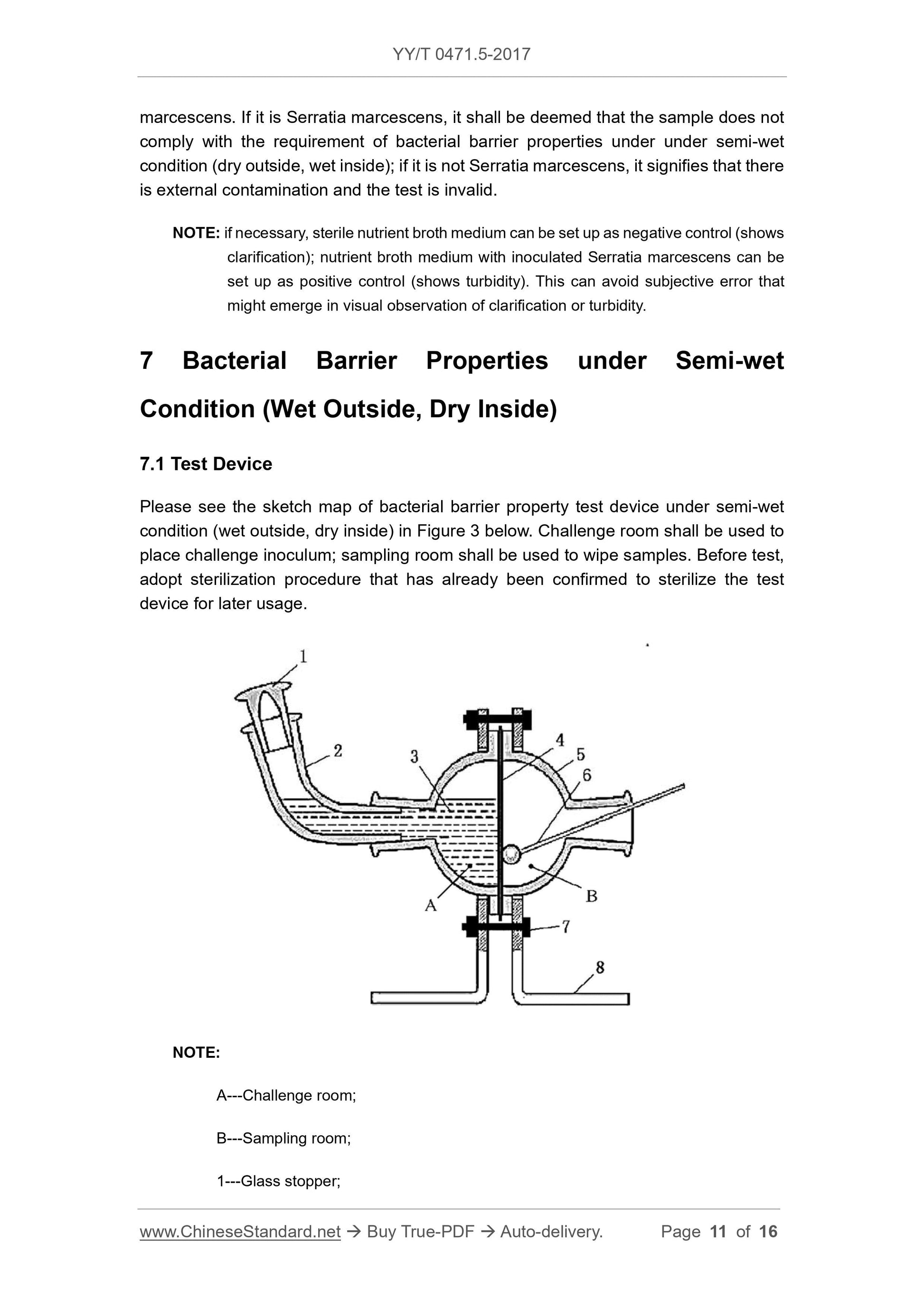
Scope
This Part of YY/T 0471 specifies test methods 1 of evaluating bacterial barrierproperties of primary wound dressings that claim to have bacterial barrier properties.
In this Part, test methods involve microbial detection, which shall be conducted by
trained personnel in biosafety laboratories.
Basic Data
| Standard ID | YY/T 0471.5-2017 (YY/T0471.5-2017) |
| Description (Translated English) | Test Methods for Primary Wound Dressings - Part 5: Bacterial Barrier Properties |
| Sector / Industry | Medical Device and Pharmaceutical Industry Standard (Recommended) |
| Classification of Chinese Standard | C48 |
| Classification of International Standard | 11.120.20 |
| Word Count Estimation | 10,128 |
| Date of Issue | 2017-02-28 |
| Date of Implementation | 2018-01-01 |
| Older Standard (superseded by this standard) | YY/T 0471.5-2004 |
| Issuing agency(ies) | State Food and Drug Administration |
Share