1
/
of
8
www.ChineseStandard.us -- Field Test Asia Pte. Ltd.
GB 29922-2013 English PDF
GB 29922-2013 English PDF
Regular price
$85.00
Regular price
Sale price
$85.00
Unit price
/
per
Shipping calculated at checkout.
Couldn't load pickup availability
GB 29922-2013: National food safety standard - General principles for formula foods of special medical purpose
Delivery: 9 seconds. Download (and Email) true-PDF + Invoice.Get Quotation: Click GB 29922-2013 (Self-service in 1-minute)
Newer / historical versions: GB 29922-2013
Preview True-PDF
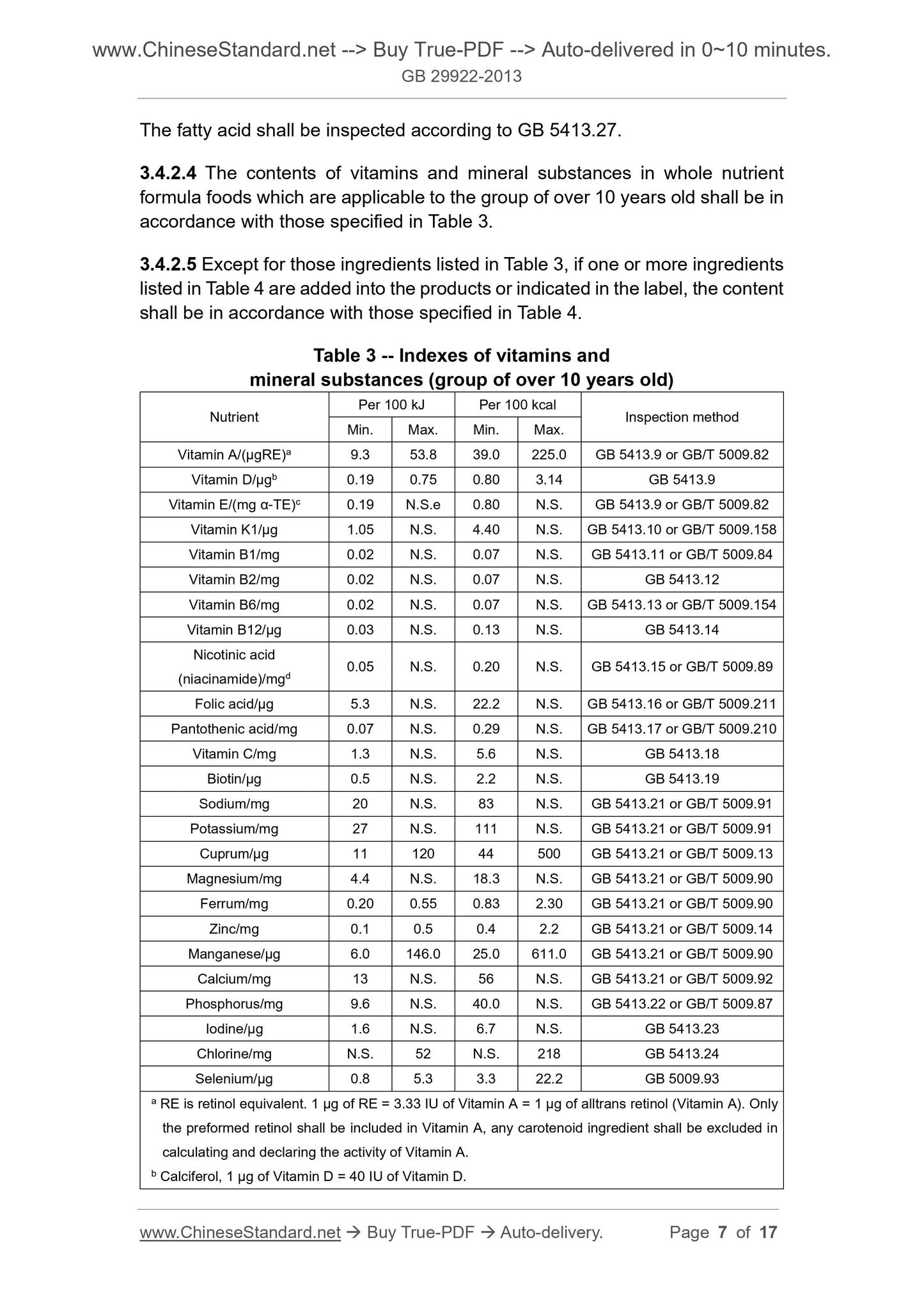
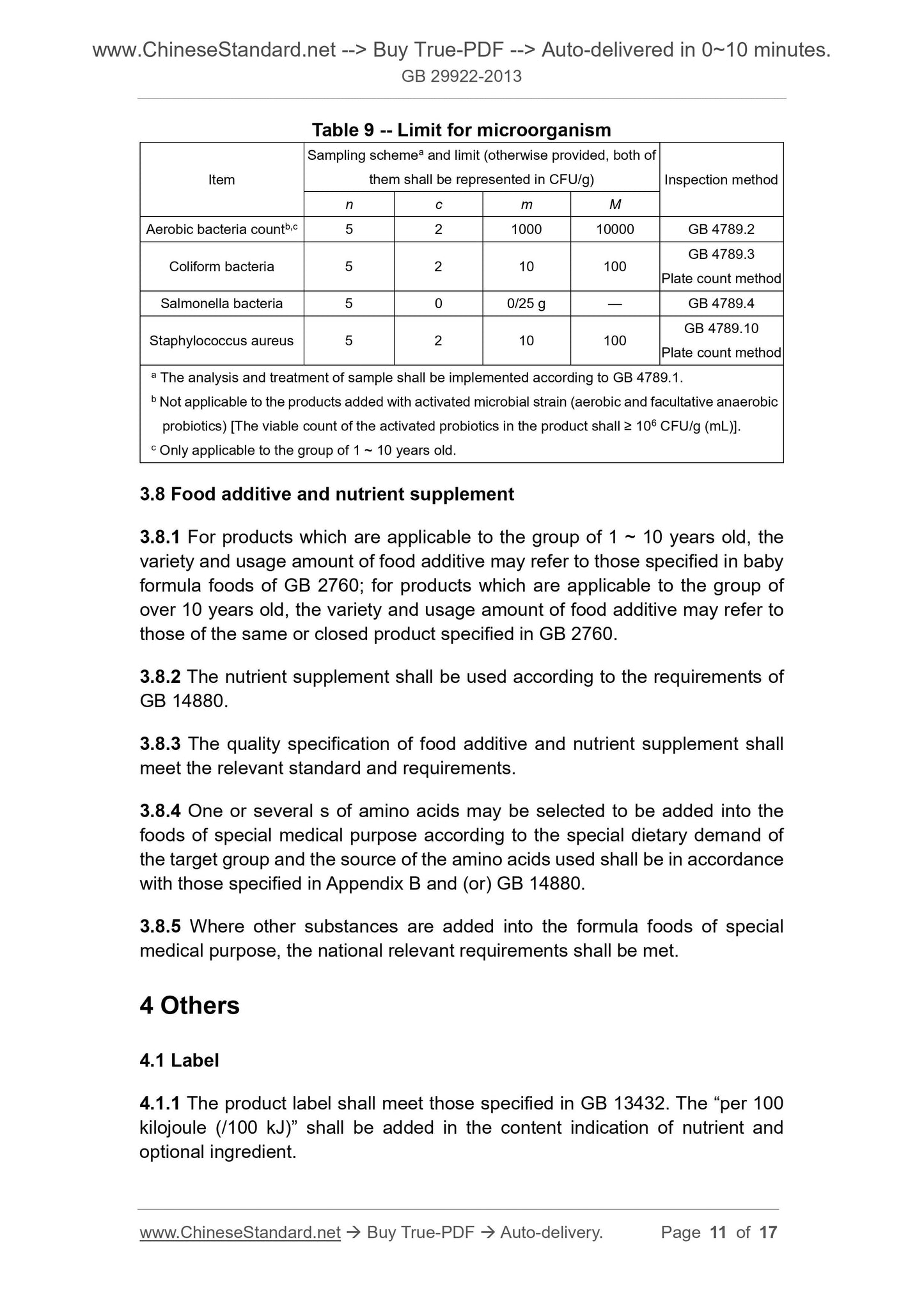
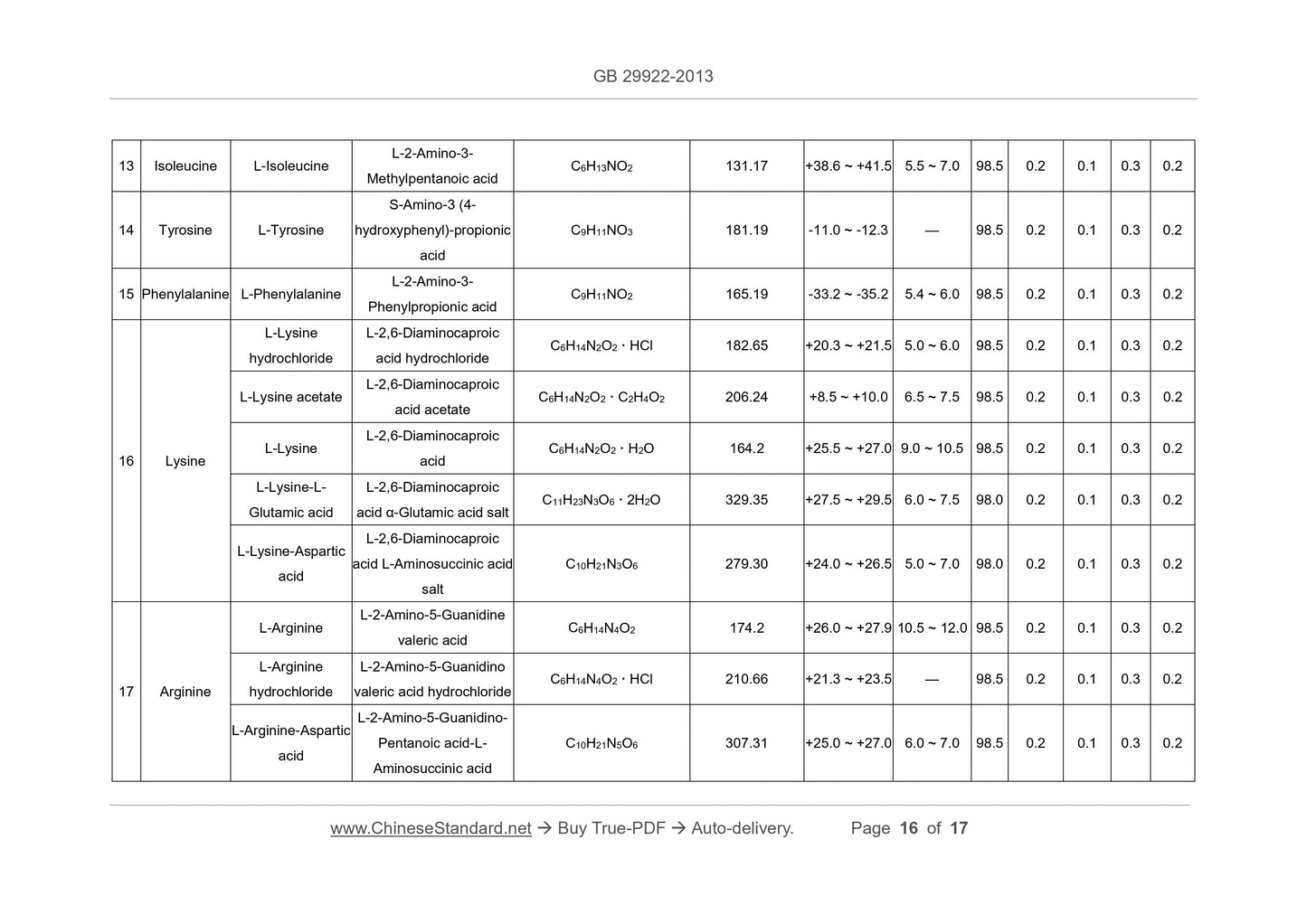
Scope
This Standard is applicable to formula foods of special medical purpose forpeople older than 1 year old.
Basic Data
| Standard ID | GB 29922-2013 (GB29922-2013) |
| Description (Translated English) | National food safety standard - General principles for formula foods of special medical purpose |
| Sector / Industry | National Standard |
| Classification of Chinese Standard | C53 |
| Classification of International Standard | 67.020 |
| Word Count Estimation | 16,174 |
| Quoted Standard | GB 14881 |
| Regulation (derived from) | China Food and Drug Administration [2013] No. 234, November, 1, 2013 |
| Issuing agency(ies) | National Health and Family Planning Commission of the People's Republic of China |
| Summary | This standard applies to 1 year old crowd recipes food for special medical purposes. |
Share